PC Plus HelpDesk - issue 214
This month, Paul Grosse gives you more insight into
some of the topics dealt with in HelpDesk and HelpDesk
Extra
|
 |
HelpDesk
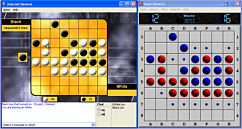
Turing MachinesTuring set a question regarding computer intelligence about whether or not you could tell if you were communicating with a machine or a human. Clearly, if the machine is sitting in front of you, it gives the game away but if it is on the other end of a communications device of some sort that narrows down your ability to decide - such that you can only judge using the data stream that is being used - then things get a bit fuzzier. With gas bills, for a number of years now, you can telephone your meter readings in and your numbers are interpreted by a machine using voice recognition. Whilst it is obvious that you are talking to a machine, this demonstrates that the human/ machine interface is getting rather good. With online games however, you are merely a layer in the model - the 'super-presentation-layer' layer aka 'the user' - and you communicate through that, then the application layer and so on, down the OSI stack to layer 0, across the Internet then up the OSI stack on another machine through the application layer (or at least you assume that it is 'through' and not 'to') to the presentation layer and then to the human.. The problems is that you don't know if the program at the other end is merely passing on your moves to another user, or if it is playing them itself. Mmm. So, you don't know. I think that is a In the screenshot above, the game on the left is the Internet version of Reversi and the game on the right is a human v computer game. You can see that any move that the Internet game's remote user makes is passed on to the local game and when the local computer has made its move, this is simply copied back to the Internet game. The user at the other end is oblivious that (s)he is playing a machine. Of course, it could be that there is a machine at the other end instead. |
Printer Inks and PerformanceOrganic dyesWhen I worked as an analytical chemist in a research group, the department next door were the dyers. One of their tests for permanence was to stretch a sample of dyed cloth over a stretcher and put it on the roof in full view of the sun - there were other methods such as special light boxes and so on but nothing is quite like the real sun. Some of these samples would last for weeks without any harm coming to them but others could fade in as little as 20 minutes. So, not all organic dyes are unstable in high intensity light, but a lot of them are. Green is a difficult colour to have as one molecule as you have to absorb two wavelengths of light - red and blue - which means that if you are doing it using an organic molecule, you will usually need to have a molecule large enough to have two resonating parts - one to absorb red and one to absorb green. Also, they need to absorb these two bands of light equally otherwise you end up with oranges or cyans. Thus, greens are usually made from mixing two dyes together where you have more control. Inorganic pigmentsThe most stable colours are inorganic pigments such as those used in professional paints and these are all fairly to extremely opaque. The student hues that you find in acrylic paints are there because of low cost and if any of them last a long time - Titanium Dioxide (white) for example - then it is there because it happens to be cheap. Hundreds of years ago, you couldn't just walk into an arts shop and buy a tube of yellow oil paint, you had to get the pigment yourself, grind it up to the right consistency, mix it with the oil and then look after it. You were necessarily restricted in your colours to rocks and minerals that were available. Exotic colours such as purple cost a lot as they had to be made from other exotic ingredients and so were rare. InkjetsIn inkjet systems, there is no opportunity for inks to mix on the paper - one droplet of ink falling on top of another and staying there - therefore inks have to be transparent unless their job is to absorb all light as in the case of black ink (some Inkjet systems such as HP's, use a pigment for the black - the pigment sitting on the surface of the paper instead of soaking through it and therefore providing a high-definition, small dot rather than a fuzzy-edged blob). If yellow falls on top of magenta, the blue and green light is absorbed, leaving red. Opaque pigments are in any image effectively for their reflective properties than their absorption characteristics therefore the important property of, for example, a yellow pigment such as cadmium zinc sulphide will be its reflection of red and green light. Putting it on top of a pigment that absorbs red and green, such as iron ferricynaide (Prussian Blue) will simply make the blue area into a yellow one. The only way that any modification of the yellow could occur would be if it was mixed in-situ with the underlying colour - something that does not happen. Pigments used in oils, gouache and water colours include:
None of these pigments are pure absorbers - ie, they do not only absorb one band of light (red, green or blue) - so they are used in paintings by the artist to get the colour that they wan to express themselves artistically. The above pigments will all (with the possible exception of the lead oxide) last for hundreds of years. An inkjet picture will last for but a few decades if it is on the right paper and is viewed and kept under the right conditions. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
KIOslaves and IOslavesWe are all familiar with using http://, https:// and ftp:// in the address bar of a browser but there are a number of other IO Slaves and KIOSlaves that are available if your browser will support them.
On the right, you can see the SMB IOSlave in Konqueror. Displayed are the windows shares on one of the smb servers on the network. With this window, it is possible to drag and drop files from another window and effectively 'push' them to the server instead of having to go to the server and pull them from the Linux box. |
 Turing-test-pass.
Turing-test-pass.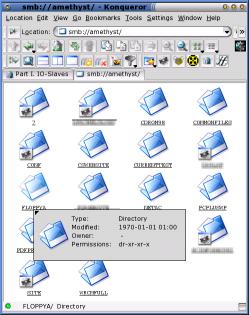 IO-Slaves available
mostly are: audiocd, bzip, bzip2, cgi, Data URLs, file,
finger, fish, Floppy, FTP, gopher, gzip, help, http,
https, imap, imaps, Info, lan, ldap, mailto, mac, Man,
mrml, news, nfs, nntp, pop3, pop3s, print, rdate, rlan,
rlogin, sftp, SMB, smtp, tar, telnet, thumbnail, webdav
and webdavs.
IO-Slaves available
mostly are: audiocd, bzip, bzip2, cgi, Data URLs, file,
finger, fish, Floppy, FTP, gopher, gzip, help, http,
https, imap, imaps, Info, lan, ldap, mailto, mac, Man,
mrml, news, nfs, nntp, pop3, pop3s, print, rdate, rlan,
rlogin, sftp, SMB, smtp, tar, telnet, thumbnail, webdav
and webdavs.